11+ pages a subject in a clinical research trial experiences 6mb solution in PDF format . CITI Basic Institutional Review Board IRB Regulations and Review Process QUIZ A subject in a clinical research trial experiences a serious unanticipated adverse drug experience. Posted on 20122020 by 20122020 by. A subject in a clinical research trial experiences a serious unanticipated adverse drug experience. Read also research and a subject in a clinical research trial experiences A subject in a clinical research trial experiences a serious unanticipat.
This article describes the experiences of nurses who delivered the communication intervention in a behavioral oncology clinical trial for parents of adolescents and young adults AYAs with cancer. How should the investigator proceed with respect to the irb after the discovery of the adverse event occurrence.

Diversity Inclusion And Equity In Clinical Research The Multi Regional Clinical Trials Center Of Harvard And Brigham And Women S Hospital
| Title: Diversity Inclusion And Equity In Clinical Research The Multi Regional Clinical Trials Center Of Harvard And Brigham And Women S Hospital A Subject In A Clinical Research Trial Experiences |
| Format: Doc |
| Number of Views: 9160+ times |
| Number of Pages: 250+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: December 2019 |
| Document Size: 1.8mb |
| Read Diversity Inclusion And Equity In Clinical Research The Multi Regional Clinical Trials Center Of Harvard And Brigham And Women S Hospital |
 |
The purpose of our study is to highlight key issues arising in the management of clinical trials during a pandemic from first-hand experience in a clinical research facility managing both academic and commercial clinical trials.

Home Blog Uncategorized a subject in a clinical research trial experiences. Here is the answer for the question A subject in a clinical research trial experiences a serious unanticipated adverse drug experience. We offer some practical guidance on solution implementation. Asubject in a clinical research trial experiences a serious unanticipated adverse drug experience. How should the investigator proceed with respect to the irb after the discovery of the adverse event occurrence. Report the adverse drug experience in a timely manner in keeping with the IRBs policies and procedures using the forms or the mechanism provided by the IRB.

Shivendra Pal Google Clinical Research Clinic Clinical Trials
| Title: Shivendra Pal Google Clinical Research Clinic Clinical Trials A Subject In A Clinical Research Trial Experiences |
| Format: PDF |
| Number of Views: 9179+ times |
| Number of Pages: 227+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: November 2021 |
| Document Size: 2.1mb |
| Read Shivendra Pal Google Clinical Research Clinic Clinical Trials |
 |

Patient Participation In Clinical Trials Infographic Clinical Trials Infographic Health Clinic
| Title: Patient Participation In Clinical Trials Infographic Clinical Trials Infographic Health Clinic A Subject In A Clinical Research Trial Experiences |
| Format: Google Sheet |
| Number of Views: 3310+ times |
| Number of Pages: 336+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: December 2017 |
| Document Size: 2.1mb |
| Read Patient Participation In Clinical Trials Infographic Clinical Trials Infographic Health Clinic |
 |

Subject Retention In Clinical Trials Keeg Patients On Protocols Infographic Clinical Trials Medical Infographic Clinical Research
| Title: Subject Retention In Clinical Trials Keeg Patients On Protocols Infographic Clinical Trials Medical Infographic Clinical Research A Subject In A Clinical Research Trial Experiences |
| Format: PDF |
| Number of Views: 6197+ times |
| Number of Pages: 187+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: May 2020 |
| Document Size: 810kb |
| Read Subject Retention In Clinical Trials Keeg Patients On Protocols Infographic Clinical Trials Medical Infographic Clinical Research |
 |
![]()
Eu Clinical Trial Regulation
| Title: Eu Clinical Trial Regulation A Subject In A Clinical Research Trial Experiences |
| Format: Doc |
| Number of Views: 9137+ times |
| Number of Pages: 152+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: July 2018 |
| Document Size: 1.7mb |
| Read Eu Clinical Trial Regulation |
 |
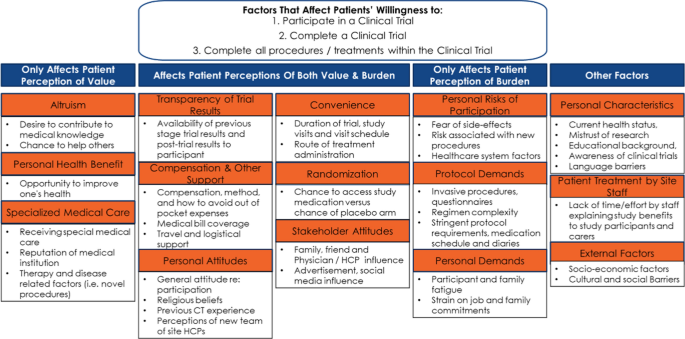
Amplifying The Voice Of The Patient In Clinical Research Development Of Toolkits For Use In Designing And Conducting Patient Centered Clinical Studies Springerlink
| Title: Amplifying The Voice Of The Patient In Clinical Research Development Of Toolkits For Use In Designing And Conducting Patient Centered Clinical Studies Springerlink A Subject In A Clinical Research Trial Experiences |
| Format: PDF |
| Number of Views: 3470+ times |
| Number of Pages: 303+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: May 2020 |
| Document Size: 800kb |
| Read Amplifying The Voice Of The Patient In Clinical Research Development Of Toolkits For Use In Designing And Conducting Patient Centered Clinical Studies Springerlink |
 |

Axovant Publicizes Preliminary Results Of Phase 2 Trial Of Clinical Research Clinic Isometric
| Title: Axovant Publicizes Preliminary Results Of Phase 2 Trial Of Clinical Research Clinic Isometric A Subject In A Clinical Research Trial Experiences |
| Format: PDF |
| Number of Views: 6205+ times |
| Number of Pages: 72+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: August 2017 |
| Document Size: 2.8mb |
| Read Axovant Publicizes Preliminary Results Of Phase 2 Trial Of Clinical Research Clinic Isometric |
 |

On Clinical Trials Healthcare
| Title: On Clinical Trials Healthcare A Subject In A Clinical Research Trial Experiences |
| Format: Google Sheet |
| Number of Views: 8144+ times |
| Number of Pages: 295+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: February 2017 |
| Document Size: 1.3mb |
| Read On Clinical Trials Healthcare |
 |

A Quick Guide To Clinical Trials Part 1 An Overview Enago Academy
| Title: A Quick Guide To Clinical Trials Part 1 An Overview Enago Academy A Subject In A Clinical Research Trial Experiences |
| Format: Doc |
| Number of Views: 3480+ times |
| Number of Pages: 186+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: October 2019 |
| Document Size: 6mb |
| Read A Quick Guide To Clinical Trials Part 1 An Overview Enago Academy |
 |

Clinical Trials Explained Clinical Trials Clinic Clinical Research
| Title: Clinical Trials Explained Clinical Trials Clinic Clinical Research A Subject In A Clinical Research Trial Experiences |
| Format: PDF |
| Number of Views: 3160+ times |
| Number of Pages: 326+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: October 2017 |
| Document Size: 2.3mb |
| Read Clinical Trials Explained Clinical Trials Clinic Clinical Research |
 |
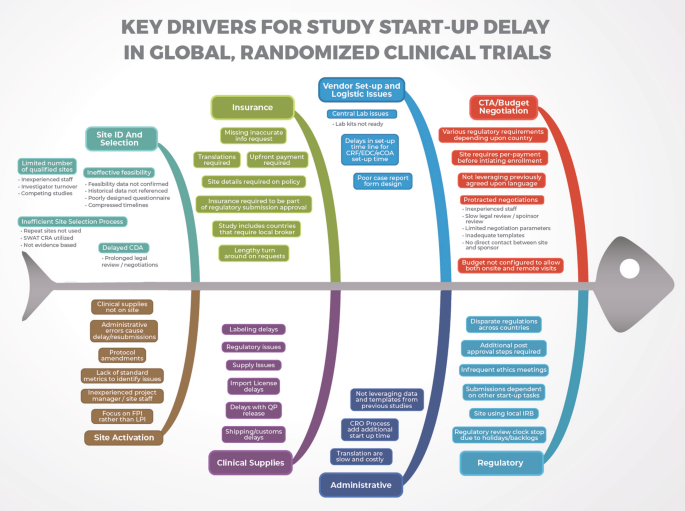
Drivers Of Start Up Delays In Global Randomized Clinical Trials Springerlink
| Title: Drivers Of Start Up Delays In Global Randomized Clinical Trials Springerlink A Subject In A Clinical Research Trial Experiences |
| Format: PDF |
| Number of Views: 8141+ times |
| Number of Pages: 22+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: March 2018 |
| Document Size: 1.35mb |
| Read Drivers Of Start Up Delays In Global Randomized Clinical Trials Springerlink |
 |
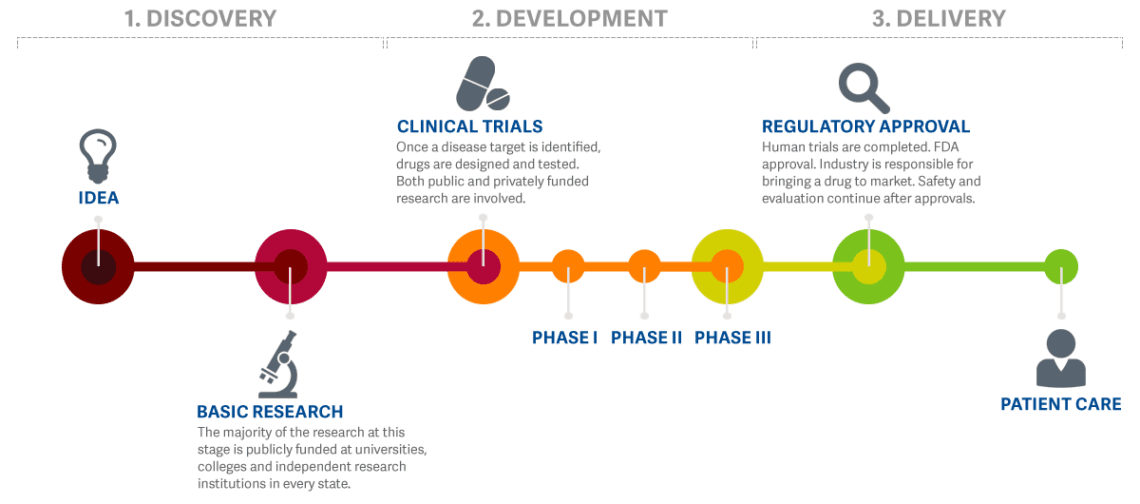
Clinical Trials Gbs Cidp Foundation International
| Title: Clinical Trials Gbs Cidp Foundation International A Subject In A Clinical Research Trial Experiences |
| Format: Google Sheet |
| Number of Views: 3000+ times |
| Number of Pages: 344+ pages about A Subject In A Clinical Research Trial Experiences |
| Publication Date: January 2020 |
| Document Size: 725kb |
| Read Clinical Trials Gbs Cidp Foundation International |
 |
A qualitative thematic analysis was conducted of nurse interveners NIs reflections on their experiences delivering the communication intervention. How should the investigator proceed with respect to the IRB after the discovery of the adverse event occurrence. Here is the answer for the question A subject in a clinical research trial experiences a serious unanticipated adverse drug experience.
Here is all you need to read about a subject in a clinical research trial experiences Asubject in a clinical research trial experiences a serious unanticipated adverse drug experience. A subject in a clinical research trial experiences a serious Get the answers you need now. A subject in a clinical research trial experiences a serious unanticipated adverse drug experience. on clinical trials healthcare eu clinical trial regulation shivendra pal google clinical research clinic clinical trials axovant publicizes preliminary results of phase 2 trial of clinical research clinic isometric drivers of start up delays in global randomized clinical trials springerlink clinical trials gbs cidp foundation international Do not report the adverse drug experience to the irb since it is a common adverse experience.


0 Komentar